[ad_1]
In 2017, Vinod Balachandran printed a paper within the science journal Nature explaining an attention-grabbing phenomenon that he had found in a tiny variety of pancreatic most cancers survivors. T-cells circulating of their blood had developed the power to establish, keep in mind and struggle again towards proteins within the lethal tumours.
The surgeon, from New York’s Memorial Sloan Kettering Most cancers Heart, likened it to “auto-vaccination”. Balachandran described how precise vaccines utilizing messenger RNA molecules may very well be used to copy the response and provides extra sufferers the power to defend themselves towards the customarily deadly tumours.
His analysis caught the attention of a then little-known scientist, Ugur Sahin, chief government of German biotechnology firm BioNTech, who was so intrigued by the findings that he invited Balachandran’s group to Mainz. Over dinner at Heiliggeist, an almost 800-year-old church-turned-restaurant on the banks of the river Rhine, and joined by scientists from Swiss pharmaceutical firm Genentech, the group mentioned the potential of mRNA vaccines to deal with pancreatic most cancers.

“It was stunning,” says Balachandran concerning the restaurant that after served as a hospital, and the dialog: “The aim and the mission was frequent between us.”
Survival charges amongst pancreatic most cancers sufferers are low. Solely 10 per cent survive longer than 5 years, in keeping with the American Most cancers Society, making it one of many deadliest types of the illness. By comparability, 90 per cent of breast most cancers sufferers survive over the identical time period.
Two years of analysis adopted the dinner and in December 2019, 20 sufferers had been enrolled within the first scientific trial assessing mRNA vaccines in pancreatic most cancers victims. With the world about to be taught of a novel coronavirus, BioNTech and others would quickly pivot their mRNA work to create a vaccine towards Covid-19.
Whereas the mRNA vaccines made by BioNTech/Pfizer and Moderna have change into synonymous with serving to to dramatically cut back deaths from Covid-19, Balachandran is amongst a rising group of scientists utilizing the medical know-how to research therapies for different diseases.
Proponents of mRNA argue that combating Covid-19 is simply the beginning and that its wider adoption heralds a revolution in fashionable medication. Cures for some types of most cancers are amongst a number of areas being explored. Pharmaceutical firms are actually turning their consideration to the ability of mRNA to deal with a spread of diseases from flu to coronary heart illness and HIV. Very early vaccine trials are additionally beneath manner for the Zika virus, yellow fever and uncommon illnesses corresponding to methylmalonic acidemia, the place the physique is unable to interrupt down proteins.
“5 years in the past there was hesitation from the bigger firms about investing on this area,” says Michael Choy, head of life sciences at Boston Consulting Group. “Having so many individuals obtain the mRNA product [for Covid] has made a giant distinction.”
Covid modifications all the things
The success of the Covid-19 vaccines has reworked the scientific and industrial view of the know-how. No mRNA-based product had ever been accredited by regulators till the disaster, and regardless of years of analysis the know-how was regarded by some within the business as troublesome to commercialise.
“It’s usually a mix between medical want and feasibility,” says Sahin, about how the corporate has chosen the diseases to focus on.
BioNTech’s focus has all the time been on creating individualised vaccines tailor-made to assault particular cancers, an method that Sahin, an oncologist, believes will revolutionise the remedy of the illness. The corporate has begun drug trials to deal with colorectal, breast, pores and skin and different cancers.
Different drugmakers together with Moderna are additionally finding out personalised most cancers vaccines utilizing mRNA. They hope to deal with illnesses which can be among the many main causes of demise worldwide whereas additionally tapping into the multibillion-dollar oncology market. Gross sales of most cancers therapeutics are forecast to hit $250bn by 2024, up from $143bn in 2019, in keeping with McKinsey.
“The motivation for this individualised most cancers vaccine is that each tumour is totally different,” Sahin says, including that even sufferers with the identical most cancers kind should not have equivalent tumours, that means a personalised remedy is prone to be simpler than a one-size-fits-all method.
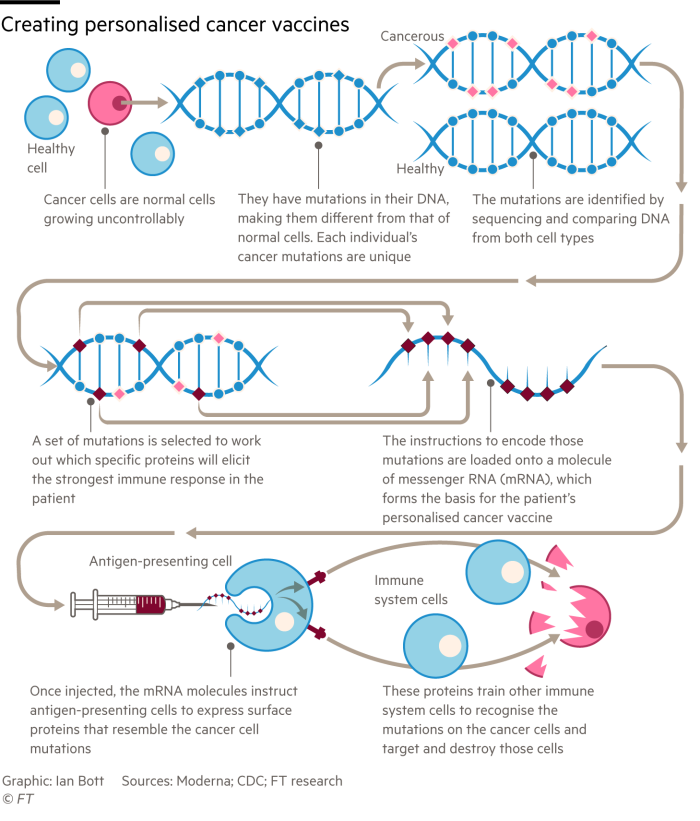
Therapeutic most cancers vaccines goal to stimulate an immune response towards current tumours, moderately than stopping illness like a flu shot. They’re tailor-made to the precise mutations in a affected person’s tumour. Scientists take away tissue from the tumour via a biopsy after which sequence the mutations discovered within the most cancers cells. The findings are in comparison with the DNA in a affected person’s blood and algorithms are used to foretell which particular proteins will elicit the strongest immune response. These proteins are then encoded — 20 of them, by each BioNTech and Moderna — into an mRNA molecule that varieties the essence of the most cancers vaccine.
As soon as injected, the directions carried on the mRNA vaccine inform the physique’s cells to specific sure proteins which practice the immune system to recognise the mutations on the most cancers cells as overseas brokers, after which assault and destroy these cells. “We began in 2014 and the time from tumour pattern to vaccine was about three months however now with automation . . . it takes lower than six weeks,” Sahin says.
Present most cancers vaccines primarily goal the virus inflicting the most cancers, moderately than the tumour itself. Within the US, the non-mRNA HPV vaccine is given to youngsters as younger as 9 to be able to shield towards cervical most cancers, which could be brought on by the human papillomavirus.
No extra ‘dabbling’
Past most cancers, mRNA trials are beneath manner for varied infectious illnesses. Influenza vaccine research are anticipated to provide outcomes most rapidly. An infectious illness corresponding to Covid or flu mutates over time and so vaccines should be up to date yearly for brand spanking new strains. Present flu vaccinations use inactivated variations of the virus and supply between 40 per cent and 60 per cent safety as a result of from the time the vaccine is made to when it’s administered, the virus has usually already mutated.
It’s hoped that mRNA, which could be tailored extra rapidly, will dramatically improve the efficacy of seasonal flu jabs. Persevering with its partnership with BioNTech, Pfizer in September began trials of an mRNA flu vaccine for adults aged between 65 and 85, one of many teams most susceptible to the sickness.
“The bottom hanging fruit is in viral vaccines as a result of we have now this clear proof of idea,” says Philip Dormitzer, chief scientific officer of Pfizer. “However we don’t suppose that’s the endpoint.”
He provides that the corporate was already working with BioNTech on creating a flu shot when Covid hit “so we clearly switched to work on a Covid-19 vaccine utilizing very a lot the know-how that we had been getting ready for the flu vaccine. As bandwidth opens up, we are actually going again to engaged on the flu vaccine.”
Pfizer’s flu jab is its solely different mRNA collaboration with BioNTech thus far. “I feel we’re able to going alone for all the things however that doesn’t essentially imply that’s what we’ll select to do,” says Dormitzer. The corporate has but to disclose which different areas it plans to focus on with mRNA however Dormitzer says uncommon illnesses, protein alternative and gene modifying “are all of curiosity”.
“There could also be firms who say ‘we have now 20 vaccines in our pipeline. You’re not going to see that method coming from Pfizer,” he provides.
In contrast, at Moderna’s annual analysis and growth day in September, the corporate laid out its mRNA plans — all 34 of them, in six totally different areas of drugs. The 11-year-old biotech group, whose inventory market ticker is MRNA, is spending about half of its power on tackling respiratory viruses and different infectious illnesses, in keeping with Stephen Hoge, its president, and the opposite half on most cancers vaccines, uncommon illnesses and gene remedy.
“It’s tragic that we’re going to have about 4m Covid deaths this 12 months,” says Hoge, “however yearly, there are about 4m deaths from respiratory viruses. The distinction is that it’s simply in smaller buckets . . . half 1,000,000 right here, 100,000 there, and it totals as much as a terrifying quantity yearly.”
The Massachusetts-based firm goals to create a pan-respiratory vaccine that would offer mixed immunity from Covid-19, flu and different infections corresponding to respiratory syncytial virus — a standard illness that may trigger lung infections — in a single jab. “No person desires to be a pin cushion,” provides Hoge. “We are able to truly get this into one needle.”
Every of Moderna’s respiratory vaccines should be individually assessed earlier than a mix is made. The corporate began trials of its flu shot in July whereas its vaccine for cytomegalovirus, a illness that has no vaccine and may trigger start defects in infants, is in part 2 trials and nonetheless a way from regulatory approval.
Responding to criticism that Moderna — whose Covid vaccine is its solely accredited drug so far — is aiming too excessive with 34 programmes, Hoge argues that though some pharmaceutical firms are “dabbling” in mRNA now that its effectiveness has been confirmed by the Covid vaccines, Moderna is all in.
Success will not be assured
The scientific and industrial success of the 2 mRNA Covid-19 vaccines has spurred a rush of funding into the sector. New mRNA therapies are anticipated to start getting into the market from 2025, in keeping with analysis by Boston Consulting Group. Revenues are anticipated to peak at $23bn in 2035, with prophylactic and therapeutic most cancers vaccines comprising 50 and 30 per cent of gross sales respectively.


Julia Angeles, funding supervisor at Baillie Gifford, an early investor in Moderna, believes that mRNA is ready to revolutionise many facets of drugs. Baillie Gifford is the largest single investor in Moderna with a 11.4 per cent stake and is the fourth largest shareholder of German mRNA-focused firm CureVac, underscoring the group’s religion in the way forward for the tactic.
“I genuinely suppose that Moderna goes to be the primary biotech firm to achieve a $1tn valuation,” says Angeles, of an organization at present valued at $124bn. “In 5 years it’s probably . . . as a result of nobody has the breadth and depth of know-how that Moderna has.”
Some would possibly dismiss that as investor hype, however different firms are priming to compete.
French pharmaceutical group Sanofi stopped trials of its personal mRNA Covid jab in October, saying that it was too late to enter a market dominated by BioNTech/Pfizer and Moderna. Nonetheless, the corporate has dedicated to the potential of the know-how by organising an mRNA centre to develop vaccines into which it’s going to make investments €400m a 12 months. Sanofi additionally purchased its accomplice Translate Bio for $3.2bn in August, hoping to capitalise on its mRNA therapeutics in areas together with cystic fibrosis and lung illness.
US drugmaker Merck can also be on the prowl for acquisitions, eyeing up a number of mRNA therapeutics firms; whereas within the UK, AstraZeneca struck its first RNA deal in September, partnering with VaxEquity to develop as much as 26 medication.
But, regardless of the optimism and Covid-era breakthroughs, it’s going to take years earlier than trials in some areas begin producing outcomes and for medication to be accredited. Regulators around the globe accelerated their approval processes throughout the pandemic due to the pressing want for a vaccine, a velocity that’s unlikely to be replicated for different medicines.
Hoge says Moderna’s respiratory syncytial virus vaccine, which is ready to maneuver to part 2 trials, may very well be prepared in three years, if the information is profitable. However he acknowledges that “the Covid pandemic was a novel circumstance”.
“If individuals need to be . . . somewhat extra conservative, or see somewhat extra information earlier than they decide, it may take just a few years. However I hope quicker than that,” he provides.
The probability of failure is extremely excessive. Lower than 10 per cent of medication that enter part 1 trials ever attain the market, in keeping with analysis by the Washington-based Biotechnology Innovation Group. Almost 60 per cent of medication which make it to part 3 trials nonetheless fail.
David Braun, an oncologist specializing in kidney most cancers on the Dana-Farber Most cancers Institute in Boston, says it’s a lengthy street from Covid vaccine to personalised most cancers jab. “Medication has made this error many instances up to now, going from enthusiasm and nice concepts to overpromising,” he says. “There’s a whole lot of promise for mRNA for use past infectious illness nevertheless it’s a giant leap.”
And mRNA vaccines don’t all the time present blockbuster outcomes. German biotech CureVac deserted its mRNA Covid vaccine on Tuesday after disappointing trial outcomes confirmed solely 48 per cent efficacy. The corporate has determined to give attention to its mRNA Covid jab with GlaxoSmithKline as an alternative. “It’s an instance that we don’t know all the things we have to know but about what makes these therapies work,” says BCG’s Choy.
Deciding on the diseases to focus on will probably be a vital resolution for brand spanking new entrants to the mRNA market.

“It doesn’t make sense to interchange, for instance, a protein-based vaccine which is very efficient, has 95 per cent effectiveness, and attempt to make an mRNA,” says BioNTech’s Sahin. “The query right here is what’s going to be improved?”
Vaccines for chickenpox, shingles and MMR are unlikely to get replaced by mRNA-based therapies as they’re efficient and researchers are specializing in diseases the place sufferers’ outcomes could be improved.
However armed with the success of the Covid vaccines the business’s high scientists are usually not quick on large and daring ambitions.
Sahin factors to the prospect of gene remedy to assist restore broken tissues and organs as a potential frontier that mRNA can assist cross within the many years to come back, probably opening the way in which to delivering new gene therapies corresponding to Crispr. “Organ restore will probably be an necessary matter for the longer term,” he says, “that is thrilling.”
[ad_2]
Source link